France Vaccines For Human Medicine Market 2026 Analysis and Forecast to 2035
Executive Summary
The French market for vaccines for human medicine occupies a position of global significance, characterized by a unique duality as both a leading consumer and the world's preeminent production hub. In 2024, France was the third-largest global consumer, with a volume of 7.7K tons, while simultaneously standing as the largest producer worldwide, with an output of 32K tons. This substantial production surplus underpins a robust export-oriented industry, though the market remains integrated within complex international supply chains, as evidenced by significant imports from key partners like Belgium and the United States. The market's financial metrics reveal a story of evolving value, with average import prices significantly higher than export prices in 2024, though both have undergone substantial corrections from historical peaks.
This report provides a comprehensive analysis of the French vaccine market, dissecting the interplay between domestic demand, industrial capacity, and international trade. It examines the foundational drivers shaping consumption, from public health policy and demographic trends to technological innovation in vaccine platforms. The analysis extends to the structure of the supply base, the competitive dynamics among global pharmaceutical leaders and specialized biotech firms, and the critical logistics of a high-value, temperature-sensitive product. The core objective is to furnish stakeholders with a data-driven, strategic understanding of the market's current state and its trajectory through to 2035.
The outlook for the French market is framed by its entrenched strengths in research, development, and manufacturing, coupled with sustained public and private investment in immunization programs. However, this trajectory will be influenced by global competitive pressures, regulatory evolution, and the ongoing need for supply chain resilience. This analysis serves as an essential tool for executives, investors, and policymakers navigating the strategic opportunities and challenges within this vital segment of the healthcare economy.
Market Overview
The French vaccine market is a cornerstone of both the national healthcare system and the global pharmaceutical industry. Its scale is underscored by its consumption of 7.7K tons in 2024, positioning it as the third-largest national market globally, behind only China and the United States. This level of consumption reflects a mature, comprehensive immunization landscape supported by strong public health infrastructure and a high degree of public acceptance. The market encompasses a broad portfolio, including routine pediatric immunizations, seasonal influenza vaccines, and advanced therapeutic vaccines for oncology and other diseases.
France's most distinctive feature is its unparalleled production capacity. With an output of 32K tons in 2024, the country accounted for approximately 33% of global vaccine production volume. This output was more than double that of the second-largest producer, the United States. This industrial dominance is not a recent phenomenon but the result of decades of strategic investment, public-private partnerships, and the presence of world-leading research clusters. The production base serves a dual purpose: fulfilling a significant portion of domestic demand and supplying a vast international market.
The market's financial dimensions are captured in its trade price points. In 2024, the average price for imported vaccines was $282,597 per ton, while the average export price was $143,227 per ton. This differential suggests a product mix variance, with France potentially importing higher-value, novel, or specialized vaccines while exporting larger volumes of established, possibly bulk-finished, products. Both price series have experienced significant volatility, having retreated from extraordinary peaks observed in the early 2010s and during the 2021 period, indicating a market in a phase of normalization and repricing following a period of disruption.
Demand Drivers and End-Use
Demand for vaccines in France is propelled by a confluence of structural, policy, and innovation-led factors. The foundational driver is the country's well-established and publicly funded immunization schedule, managed by the Ministry of Health. This schedule mandates vaccinations against a range of diseases for children and recommends others for specific age groups and at-risk populations, creating a stable, predictable baseline demand. Public trust in vaccination, though periodically challenged, remains relatively high, supported by a strong healthcare provider network that facilitates vaccine administration.
Demographic trends play a critical role in shaping demand dynamics. An aging population increases the addressable market for vaccines targeting older adults, such as those for shingles, pneumococcal disease, and high-dose influenza. Furthermore, epidemiological surveillance and the emergence of new pathogens necessitate rapid development and deployment of new vaccines, as witnessed globally. Public health campaigns, such as those promoting seasonal influenza vaccination or catch-up campaigns for HPV, directly stimulate periodic demand surges.
The end-use landscape is segmented across several key channels:
- Public Immunization Programs: The largest channel, encompassing vaccines procured by the state for the routine schedule and specific campaigns, distributed through public health centers and reimbursed for administration by private practitioners.
- Hospital and Clinic Use: Includes vaccines administered in hospital settings for inpatients, travelers, and occupational health, as well as specialized therapeutic vaccines in oncology.
- Retail Pharmacy Channel: Covers vaccines available for direct purchase or administration in pharmacies, such as certain travel vaccines, though this is a smaller segment compared to state-driven channels.
- Private Corporate Health: Occupational health programs, particularly in large industries, drive demand for vaccines against influenza and other work-related health risks.
Technological advancement is a potent demand catalyst. The successful deployment of mRNA platforms has not only expanded the arsenal against COVID-19 but has also validated a pathway for rapid development against other infectious diseases and cancers. The pipeline of next-generation vaccines, including those with broader strain coverage, improved thermostability, and novel delivery mechanisms (e.g., patch-based), promises to expand indications, improve compliance, and create new market segments, thereby sustaining long-term demand growth.
Supply and Production
France's vaccine supply landscape is dominated by its massive domestic production capability, which firmly establishes the country as the global manufacturing leader. The 2024 production volume of 32K tons, representing one-third of the world's output, is concentrated in large-scale industrial facilities operated by multinational corporations. This capacity is the result of sustained capital investment, a highly skilled workforce, and a regulatory environment that supports complex pharmaceutical manufacturing. The production ecosystem includes bulk antigen manufacturing, formulation, fill-and-finish operations, and stringent quality control laboratories.
The supply chain for vaccine production is intricate and globalized, even for a producer of France's scale. It relies on a network of suppliers for critical inputs such as:
- Specialized bioreactors and single-use technologies for cell culture-based vaccines.
- High-purity chemicals, adjuvants, and stabilizers.
- Vials, syringes, and other primary packaging materials, often subject to strict sterility and compatibility standards.
- Complex biological starting materials like cell lines and seed viruses.
Ensuring the resilience and security of this supply chain is a paramount strategic concern for producers and public health authorities alike. Recent global events have underscored the risks of over-concentration and geopolitical disruptions, prompting initiatives for regionalizing certain supply chain segments and building strategic stockpiles of key components. Production in France is characterized by high fixed costs and significant economies of scale, making capacity utilization a critical factor for profitability. The industry is also navigating a transition towards more flexible, modular production technologies that can allow for faster product switching and smaller batch sizes for niche vaccines.
Trade and Logistics
International trade is a defining feature of the French vaccine market, reflecting its role as a net exporter and a participant in global specialization. France's export profile is vast, with products reaching markets worldwide. In value terms, the leading destinations for French vaccine exports in 2024 were Belgium ($846M), Germany ($477M), and Mexico ($151M), which together accounted for a 35% share of total export value. This trade is facilitated by France's central geographic location in Europe, advanced logistics infrastructure, and the global commercial networks of the pharmaceutical majors headquartered or operating within its borders.
Conversely, France is also a significant importer of vaccines, highlighting the specialized nature of the global market. In 2024, Belgium was the leading supplier, providing $764M worth of vaccines and constituting 52% of France's total import value. The United States followed with a 13% share ($199M), and the Netherlands with 11%. This import activity can be attributed to several factors: the procurement of specific vaccines not produced domestically, intra-company transfers within multinational corporations optimizing their global manufacturing networks, and the import of novel vaccines shortly after global launch before potential local production is ramped up.
The logistics of vaccine trade are exceptionally demanding due to the products' sensitivity. The cold chain—a temperature-controlled supply chain—is non-negotiable for most vaccines, requiring:
- Specialized refrigerated storage facilities (2-8°C, -20°C, or ultra-cold -70°C).
- Validated temperature-controlled packaging for transportation.
- Real-time temperature monitoring and data loggers throughout the journey.
- Expedited customs clearance procedures to minimize transit time.
Any break in the cold chain can render a batch useless, leading to significant financial loss and potential supply shortages. Consequently, trade flows are heavily dependent on reliable air freight capacity and sophisticated logistics service providers with pharmaceutical-grade expertise. The efficiency and integrity of these logistics networks are a critical competitive advantage for France's export-oriented industry.
Price Dynamics
Price formation in the French vaccine market is influenced by a multifaceted set of factors, leading to the distinct differentials observed between import and export prices. The average import price of $282,597 per ton in 2024, though down significantly from previous highs, remained approximately double the average export price of $143,227 per ton. This disparity is not indicative of a simple quality gradient but reflects deeper structural and product-mix realities. Imported vaccines often include newer, patented products with higher development costs, such as mRNA vaccines, advanced recombinant vaccines, or combination vaccines, which command premium pricing.
The historical price data reveals a market that has experienced extreme volatility. Export prices peaked at $696,665 per ton in 2012, while import prices reached an astonishing $3,957,183 per ton in 2021. The subsequent sharp corrections, with export prices falling by -53.2% and import prices by -75.1% in 2024 alone, point to a normalization phase. Key drivers of this volatility and the prevailing price levels include:
- Product Lifecycle: Prices are highest during the patent-protected monopoly phase and decline as products mature, face competition from biosimilars (where applicable), and are incorporated into cost-constrained public tender processes.
- Procurement Mechanisms: Bulk purchasing by national health authorities through competitive tenders exerts significant downward pressure on prices for established vaccines, a dynamic strongly at play in the European market.
- Production Scale and Yield: Improvements in manufacturing efficiency and scale for high-volume products reduce unit costs, influencing export pricing.
- R&D Intensity: The price of a novel vaccine must amortize the substantial costs of clinical development, which can exceed billions of dollars, and the risk of failure.
Looking forward, price dynamics will continue to be shaped by the tension between innovation-driven value pricing and payer pressure for cost containment. The entry of new competitors in established segments, the outcomes of health technology assessments (HTAs), and global initiatives for equitable vaccine pricing will be critical factors influencing the price trajectory through the forecast period to 2035.
Competitive Landscape
The competitive environment of the French vaccine market is an oligopoly dominated by a handful of global pharmaceutical giants, complemented by a layer of innovative biotechnology firms and research institutions. The presence of Sanofi Pasteur, the vaccine division of Sanofi, headquartered in France, is the most significant factor. As one of the world's largest vaccine companies, its extensive French manufacturing and R&D footprint directly shapes the market's supply, export potential, and innovation pipeline. Other multinational players, such as GSK (GlaxoSmithKline), Pfizer, and Merck & Co., maintain substantial commercial operations, manufacturing sites, or strategic partnerships in France.
Competition operates on several parallel fronts: technological innovation, portfolio breadth, manufacturing reliability, and commercial execution. Key competitive strategies observed in the market include:
- R&D Investment: Heavy and sustained investment in discovering and developing new vaccine candidates, particularly in high-need areas like respiratory syncytial virus (RSV), universal influenza, and cancer immunotherapy.
- Portfolio Expansion: Acquiring or in-licensing promising candidates from biotech firms to fill pipeline gaps.
- Manufacturing Excellence: Competing on the reliability, scale, and speed of production, which are critical for securing large public contracts and responding to pandemic threats.
- Market Access and Partnerships: Building strong relationships with public health authorities, healthcare providers, and international organizations to ensure inclusion in immunization programs.
The competitive landscape is also influenced by public entities. The French National Institute of Health and Medical Research (Inserm), the Pasteur Institute, and various university hospitals are powerhouses of basic and translational vaccine research. They often act as innovation feeders, conducting early-stage research that is later licensed to or co-developed with industry partners. This public-private ecosystem is a key strength, ensuring France remains at the forefront of vaccine science. Furthermore, competition is increasingly global, with manufacturers in emerging markets building capacity and competing on price in certain segments, adding another layer of complexity to the strategic calculus for incumbents.
Methodology and Data Notes
This market analysis is constructed using a robust, multi-layered methodology designed to ensure accuracy, relevance, and strategic depth. The core of the analysis is based on official statistical data, including trade figures from customs authorities, production statistics from national industrial surveys, and consumption data derived from the reconciliation of production, trade, and inventory changes. This quantitative foundation is triangulated with qualitative insights from industry reports, scientific publications, company financial disclosures, and policy documents to provide context and narrative.
The forecast perspective through 2035 is developed using a combination of quantitative modeling and scenario-based qualitative analysis. Time-series analysis of historical data identifies underlying trends, growth rates, and cyclical patterns. These are then integrated with an assessment of identified market drivers and restraints—such as demographic projections, pipeline product launch probabilities, regulatory changes, and macroeconomic factors—to build a coherent forward-looking view. The analysis explicitly avoids inventing new absolute forecast figures, instead focusing on directional trends, relative shifts, and the interplay of market forces.
It is crucial to note the specific context of the data points cited. The consumption and production volumes are expressed in tons (K tons), a metric that reflects the physical volume of vaccine product. This can be influenced by the formulation (liquid vs. lyophilized) and packaging, and does not directly correlate with doses. The trade values are in nominal U.S. dollars, and prices are per ton. The year 2024 serves as the latest complete data year for the historical analysis. Readers should be aware that the vaccine market can be subject to acute, non-recurring events (e.g., pandemic response) that cause significant short-term deviations from long-term trends, and such events are factored into the analysis as singular shocks rather than new baselines.
Outlook and Implications
The French market for human vaccines is poised for evolution rather than revolution over the forecast period to 2035, building upon its entrenched strengths while navigating a set of persistent challenges. The foundational outlook is one of stable growth in domestic consumption, driven by an aging population, the expansion of immunization recommendations to new age groups and indications, and the successful launch of next-generation products from industry pipelines. France's position as the global production leader is expected to remain secure, supported by ongoing investment in modernizing manufacturing assets and adopting advanced technologies like continuous manufacturing and Industry 4.0 principles.
Several key implications for industry stakeholders emerge from this analysis. For manufacturers, the imperative will be to balance investment in groundbreaking innovation (e.g., mRNA, vector-based platforms) with the need for operational excellence and cost competitiveness in mature product segments. The ability to demonstrate not only clinical efficacy but also cost-effectiveness and real-world value will be increasingly critical for market access and favorable pricing in negotiations with French and European health authorities. Supply chain resilience will move from a tactical concern to a core strategic capability, necessitating diversification of suppliers, increased inventory buffers for critical components, and potential nearshoring of certain production steps.
For policymakers and public health officials, the outlook underscores the strategic asset represented by the domestic vaccine industry. Policies that support R&D, streamline regulatory pathways for advanced manufacturing, and foster a skilled workforce will be vital to maintaining France's competitive edge. Simultaneously, ensuring sustainable financing for the immunization program to incorporate new, often higher-priced vaccines will be an ongoing challenge. The trade landscape will continue to reflect France's dual role, with exports serving as a major economic contributor and imports ensuring rapid access to global innovation. Ultimately, the trajectory of the French vaccine market through 2035 will be a testament to the successful integration of scientific ambition, industrial prowess, and public health commitment within a dynamic global framework.
Frequently Asked Questions (FAQ) :
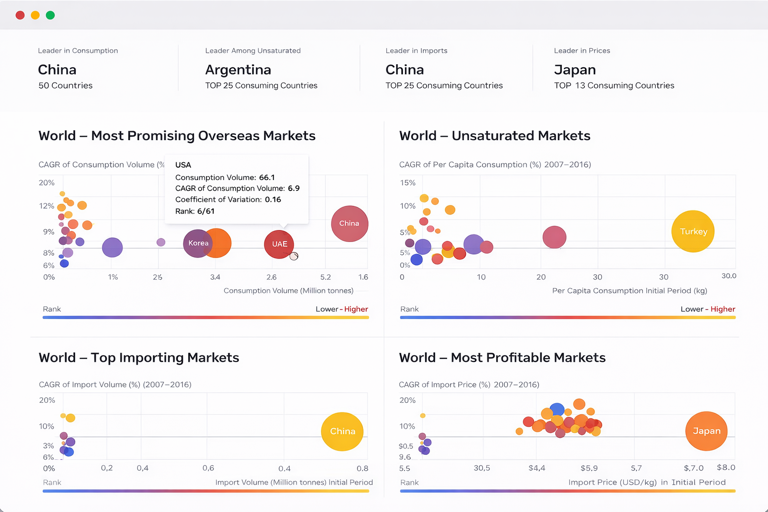
The countries with the highest volumes of consumption in 2024 were China, the United States and France, with a combined 35% share of global consumption. India, Poland, Spain, Germany, Japan, Russia and Indonesia lagged somewhat behind, together comprising a further 27%.
France remains the largest vaccine producing country worldwide, accounting for 33% of total volume. Moreover, vaccine production in France exceeded the figures recorded by the second-largest producer, the United States, twofold. The third position in this ranking was held by China, with a 13% share.
In value terms, Belgium constituted the largest supplier of vaccines for human medicine to France, comprising 52% of total imports. The second position in the ranking was taken by the United States, with a 13% share of total imports. It was followed by the Netherlands, with an 11% share.
In value terms, Belgium, Germany and Mexico appeared to be the largest markets for vaccine exported from France worldwide, with a combined 35% share of total exports. Italy, Spain, the UK and Ireland lagged somewhat behind, together comprising a further 6.6%.
In 2024, the average vaccine export price amounted to $143,227 per ton, waning by -53.2% against the previous year. Over the period under review, the export price faced a abrupt descent. The growth pace was the most rapid in 2021 an increase of 564%. Over the period under review, the average export prices attained the maximum at $696,665 per ton in 2012; however, from 2013 to 2024, the export prices failed to regain momentum.
In 2024, the average vaccine import price amounted to $282,597 per ton, which is down by -75.1% against the previous year. Over the period under review, the import price faced a abrupt descent. The most prominent rate of growth was recorded in 2021 an increase of 282% against the previous year. As a result, import price attained the peak level of $3,957,183 per ton. From 2022 to 2024, the average import prices remained at a lower figure.
This report provides a comprehensive view of the vaccines industry in France, tracking demand, supply, and trade flows across the national value chain. It explains how demand across key channels and end-use segments shapes consumption patterns, while also mapping the role of input availability, production efficiency, and regulatory standards on supply.
Beyond headline metrics, the study benchmarks prices, margins, and trade routes so you can see where value is created and how it moves between domestic suppliers and international partners. The analysis is designed to support strategic planning, market entry, portfolio prioritization, and risk management in the vaccines landscape in France.
Quick navigation
Key findings
- Domestic demand is shaped by both household and industrial usage, with trade flows linking local supply to imports and exports.
- Pricing dynamics reflect unit values, freight costs, exchange rates, and regulatory shifts that affect sourcing decisions.
- Supply depends on input availability and production efficiency, creating a distinct national cost curve.
- Market concentration varies by segment, creating different competitive landscapes and entry barriers.
- The 2035 outlook highlights where capacity investment and demand growth are most aligned within the country.
Report scope
The report combines market sizing with trade intelligence and price analytics for France. It covers both historical performance and the forward outlook to 2035, allowing you to compare cycles, structural shifts, and policy impacts.
- Market size and growth in value and volume terms
- Consumption structure by end-use segments
- Production capacity, output, and cost dynamics
- Trade flows, exporters, importers, and balances
- Price benchmarks, unit values, and margin signals
- Competitive context and market entry conditions
Product coverage
- Prodcom 21202145 - Vaccines for human medicine
Country coverage
Country profile and benchmarks
This report provides a consistent view of market size, trade balance, prices, and per-capita indicators for France. The profile highlights demand structure and trade position, enabling benchmarking against regional and global peers.
Methodology
The analysis is built on a multi-source framework that combines official statistics, trade records, company disclosures, and expert validation. Data are standardized, reconciled, and cross-checked to ensure consistency across time series.
- International trade data (exports, imports, and mirror statistics)
- National production and consumption statistics
- Company-level information from financial filings and public releases
- Price series and unit value benchmarks
- Analyst review, outlier checks, and time-series validation
All data are normalized to a common product definition and mapped to a consistent set of codes. This ensures that comparisons across time are aligned and actionable.
Forecasts to 2035
The forecast horizon extends to 2035 and is based on a structured model that links vaccines demand and supply to macroeconomic indicators, trade patterns, and sector-specific drivers. The model captures both cyclical and structural factors and reflects known policy and technology shifts in France.
- Historical baseline: 2012-2025
- Forecast horizon: 2026-2035
- Scenario-based sensitivity to income growth, substitution, and regulation
- Capacity and investment outlook for major producing companies
Each projection is built from national historical patterns and the broader regional context, allowing the report to show where growth is concentrated and where risks are elevated.
Price analysis and trade dynamics
Prices are analyzed in detail, including export and import unit values, regional spreads, and changes in trade costs. The report highlights how seasonality, freight rates, exchange rates, and supply disruptions influence pricing and margins.
- Price benchmarks by country and sub-region
- Export and import unit value trends
- Seasonality and calendar effects in trade flows
- Price outlook to 2035 under baseline assumptions
Profiles of market participants
Key producers, exporters, and distributors are profiled with a focus on their operational scale, geographic footprint, product mix, and market positioning. This helps identify competitive pressure points, partnership opportunities, and routes to differentiation.
- Business focus and production capabilities
- Geographic reach and distribution networks
- Cost structure and pricing strategy indicators
- Compliance, certification, and sustainability context
How to use this report
- Quantify domestic demand and identify the most attractive segments
- Evaluate export opportunities and prioritize target destinations
- Track price dynamics and protect margins
- Benchmark performance against leading competitors
- Build evidence-based forecasts for investment decisions
This report is designed for manufacturers, distributors, importers, wholesalers, investors, and advisors who need a clear, data-driven picture of vaccines dynamics in France.
FAQ
What is included in the vaccines market in France?
The market size aggregates consumption and trade data, presented in both value and volume terms.
How are the forecasts to 2035 built?
The projections combine historical trends with macroeconomic indicators, trade dynamics, and sector-specific drivers.
Does the report cover prices and margins?
Yes, it includes export and import unit values, regional spreads, and a pricing outlook to 2035.
Which benchmarks are included?
The report benchmarks market size, trade balance, prices, and per-capita indicators for France.
Can this report support market entry decisions?
Yes, it highlights demand hotspots, trade routes, pricing trends, and competitive context.