China Vaccines For Human Medicine Market 2026 Analysis and Forecast to 2035
Executive Summary
The Chinese market for vaccines for human medicine represents a critical and dynamic component of the global pharmaceutical landscape. As of 2024, China stands as the world's largest consumer by volume, with demand reaching 13,000 tons, yet it ranks as the third-largest global producer, manufacturing 12,000 tons. This structural gap between domestic consumption and production capacity underscores a market characterized by significant import dependency, particularly for high-value, innovative products. The market's trajectory is shaped by powerful demographic forces, proactive public health policies, and a concerted national drive toward biopharmaceutical self-sufficiency.
This report provides a comprehensive analysis of the market's current state, anchored in 2024-2025 data, and projects the strategic environment through 2035. It examines the complex interplay of demand drivers, including an aging population and expanding National Immunization Program (NIP), against the evolving domestic supply landscape, where local innovation is accelerating. The analysis further dissects the intricate trade dynamics, where high-value imports from Ireland and the United States contrast with China's export profile focused on emerging markets. The competitive arena is becoming increasingly bifurcated, featuring multinational corporations (MNCs) and a growing cohort of capable domestic champions.
The outlook to 2035 suggests a market in transition, moving from volume-based consumption towards value-driven growth. While imports will remain crucial for advanced therapies, the share of domestically produced vaccines, particularly in novel categories like mRNA and recombinant proteins, is poised to expand significantly. This evolution presents both challenges and opportunities for stakeholders across the value chain, from global suppliers to local manufacturers and policymakers. The following sections provide the detailed, data-driven foundation necessary to navigate this complex and vital market.
Market Overview
The Chinese vaccine market occupies a unique and pivotal position globally, defined by its massive scale and its ongoing structural evolution. In consumption terms, China is the undisputed global leader, with a volume of 13,000 tons in 2024, substantially ahead of the United States (9,700 tons) and France (7,700 tons). This consumption accounted for a significant portion of global demand, highlighting the critical role of China's public health infrastructure and its population of over 1.4 billion people. The sheer volume of doses administered annually through both public and private channels makes China a bellwether for global vaccine utilization trends.
However, the production landscape tells a different story. Global vaccine manufacturing is heavily concentrated, with France leading as the largest producer at 32,000 tons (approximately 33% of global output), followed by the United States at 13,000 tons. China ranks third, with a production volume of 12,000 tons, representing a 13% share of worldwide production. This indicates that while China's manufacturing base is substantial and ranks among the top globally, its output in 2024 was slightly below its domestic consumption needs on a volumetric basis. This gap is a fundamental characteristic of the market, driving trade flows and investment in capacity expansion.
The market's value dynamics reveal an even more pronounced disparity. The average import price for vaccines into China in 2024 was $3,744,281 per ton, significantly higher than the average export price of $1,367,562 per ton. This stark contrast underscores the high-value, innovative nature of imported products—such as novel pediatric combinations, HPV, and shingles vaccines—compared to the more traditional, essential vaccines that constitute a larger share of China's exports. The market is thus segmented into a high-value import corridor and a volume-driven domestic and export manufacturing sector, though this dichotomy is gradually blurring.
Regulatory oversight by the National Medical Products Administration (NMPA) has undergone significant modernization, aligning more closely with international standards through the implementation of a marketing authorization holder (MAH) system and accelerated review pathways for innovative and clinically urgent vaccines. This regulatory evolution, coupled with the "Healthy China 2030" blueprint, provides a stable yet ambitious framework for market development. The interplay between scale, value, and regulatory advancement defines a market that is both a colossal consumption engine and an increasingly sophisticated production and innovation hub.
Demand Drivers and End-Use
Demand for vaccines in China is propelled by a confluence of powerful, long-term demographic, epidemiological, and policy factors. The foundational driver is the expansive National Immunization Program (NIP), which provides free, mandatory vaccines against 14 diseases to the nation's children. This program ensures high coverage rates for essential vaccines and forms the stable, volume-based core of market demand. Beyond the NIP, the growing adoption of non-NIP, or Category II, vaccines—which are voluntarily purchased—represents the primary engine for value growth, as families and individuals increasingly seek enhanced protection against a broader range of diseases.
Demographic shifts are creating sustained demand across multiple cohorts. While the annual number of newborns has declined from previous peaks, the sheer size of the pediatric population ensures a consistent baseline demand for routine immunization. More significantly, the rapid aging of the population is catalyzing the adult and elderly vaccination segment. Increased public awareness and government advocacy for vaccines against influenza, pneumococcal disease, and herpes zoster are transforming this segment from a niche market into a major growth pillar. The epidemiological transition, with a rising burden of chronic diseases and cancers, is also fueling demand for prophylactic vaccines like HPV.
Public health policy and infrastructure development serve as critical demand enablers. The "Healthy China 2030" initiative explicitly prioritizes disease prevention, with vaccination as a cornerstone. Government-led education campaigns, often amplified through digital health platforms, have significantly improved public understanding of vaccine benefits. Furthermore, the integration of vaccination services into primary healthcare networks and the expansion of cold chain logistics across even remote regions have dramatically improved access. Periodic outbreaks of infectious diseases, as witnessed globally, also reinforce public and governmental commitment to robust immunization coverage, leading to demand spikes for relevant vaccines.
The end-use segmentation of the market is clearly stratified. The public sector, procuring for the NIP, is the dominant channel by volume but is highly price-sensitive. The private sector, including private hospitals, clinics, and standalone vaccination centers, drives the premium and innovative vaccine segment, characterized by higher margins and more diverse product offerings. This bifurcation requires suppliers to develop distinct market access, pricing, and distribution strategies for each channel. The ongoing expansion of private health insurance and corporate wellness programs is further formalizing and financing the demand for non-NIP vaccines, adding another layer of stability to this high-growth segment.
Supply and Production
China's domestic vaccine supply landscape is in a state of accelerated transformation, moving from a historical focus on traditional, inactivated vaccines towards a more diversified and innovative portfolio. The production volume of 12,000 tons in 2024, representing 13% of global output, is concentrated among a mix of large, state-affiliated enterprises and an emerging group of dynamic, science-driven biotech firms. Traditional manufacturers, such as China National Biotec Group (CNBG), dominate the supply for the NIP, producing vast quantities of vaccines for diseases like hepatitis B, polio, and measles, mumps, and rubella (MMR). Their operations are characterized by high-volume, cost-efficient production with deeply entrenched relationships in the public procurement system.
The most significant shift in supply is occurring in the innovative vaccine segment. Domestic companies are making substantial investments in advanced technology platforms, including recombinant protein expression, viral vectors, and mRNA. This is leading to a new generation of locally developed products for HPV, pneumococcal disease, herpes zoster, and respiratory syncytial virus (RSV). The success of these products, such as domestically developed HPV vaccines, demonstrates the growing technical capability and regulatory agility of China's biopharmaceutical sector. Capacity expansion for these novel vaccines is a key focus, with numerous new manufacturing facilities receiving GMP certification to international standards.
The supply chain for vaccine production is also undergoing localization and strengthening. While some critical raw materials and cell culture media may still be imported, there is a strong push for supply chain security and indigenization. This extends to the fill-and-finish stage and the vital cold chain logistics required for distribution. The government supports this through industrial policy and funding for national biomedicine parks, which cluster R&D, manufacturing, and supporting services. However, challenges remain, including the need for continuous process optimization to achieve the yield and consistency levels of established global leaders, and the management of complex global supply chains for certain advanced components.
Looking ahead, the domestic supply trajectory points towards increasing self-sufficiency in several key vaccine categories. However, this does not imply a move towards isolation. Instead, the market is evolving towards a more balanced structure where domestic innovators capture a growing share of the mid-to-high-value market, while multinational corporations (MNCs) continue to lead in the most complex and novel therapeutic areas. This dynamic will foster both competition and potential collaboration, such as co-development and licensing deals, shaping a more mature and globally integrated supply ecosystem within China by 2035.
Trade and Logistics
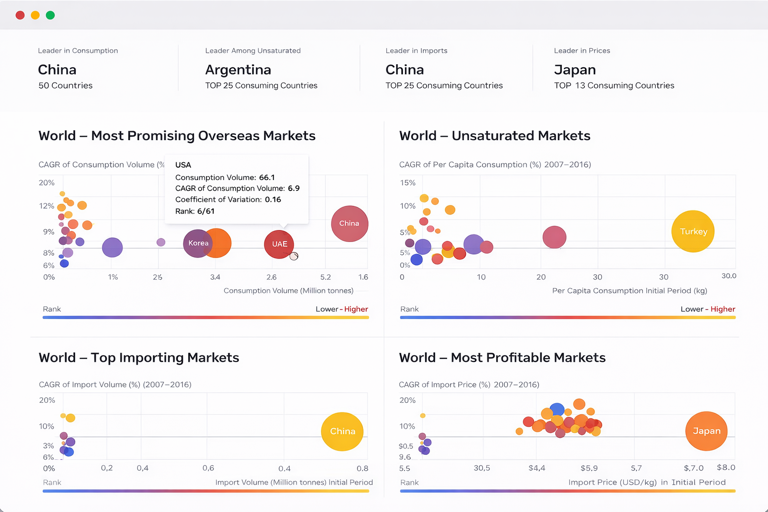
International trade is a defining feature of the Chinese vaccine market, vividly illustrating the gap between the types of vaccines demanded and those supplied domestically. China is a net importer of vaccines by value, with imports dominated by high-price, innovative products. In value terms, Ireland constituted the largest supplier in 2024, providing $3.4 billion worth of vaccines and capturing a commanding 70% share of total import value. This is followed by the United States ($589 million, 12% share) and France (9.1% share). The concentration from Ireland, a hub for several major multinational pharmaceutical companies, underscores the reliance on Western-developed novel vaccines and advanced biologics.
On the export side, China's profile is markedly different, focusing on more affordable, essential vaccines and serving primarily emerging and developing economies. In 2024, the leading destinations for Chinese vaccine exports by value were Morocco ($48 million, 22% share), Pakistan ($20 million, 9.5% share), and Egypt (8.1% share). This export pattern aligns with China's broader public health diplomacy and the capabilities of its established manufacturers to produce WHO-prequalified vaccines at scale. The exports play a crucial role in global health equity by supplying life-saving products to markets with similar disease burdens and budget constraints.
The price differential between imports and exports is the most telling trade metric. The average import price in 2024 was $3,744,281 per ton, reflecting the premium nature of imported goods. In contrast, the average export price was $1,367,562 per ton. This disparity of nearly threefold highlights the value hierarchy in the global vaccine market, which China's trade flows currently mirror. It is important to note that the export price has shown a resilient increase over recent years, growing 9.4% in 2024, suggesting a gradual move towards somewhat higher-value exported products as domestic innovation bears fruit.
Logistics and regulatory compliance for vaccine trade are exceptionally complex, governed by stringent cold chain requirements and customs procedures for biologics. The import process involves rigorous batch-by-batch release testing by the National Institutes for Food and Drug Control (NIFDC), which can impact lead times. Domestically, a vast and increasingly sophisticated cold chain network, supported by digital temperature monitoring, ensures integrity from manufacturer to vaccination point. As domestic production of temperature-sensitive mRNA and other advanced vaccines increases, the demands on this logistics infrastructure will intensify, requiring ongoing investment in reliability, capacity, and last-mile delivery solutions, particularly in lower-tier cities and rural areas.
Price Dynamics
Price formation in the Chinese vaccine market operates under a dual-track system, sharply divided between the publicly procured NIP segment and the private, voluntary (Category II) market. For NIP vaccines, prices are determined through a centralized, volume-based tender procurement process organized by provincial governments. This mechanism exerts intense downward pressure on prices, favoring manufacturers with the lowest costs and largest scale. Prices in this segment are relatively stable and low, reflecting their status as public health goods, and are subject to government price caps or guidance. The focus is overwhelmingly on affordability and universal access, with minimal room for premium pricing.
In the private Category II market, price dynamics are more aligned with global pharmaceutical norms, though with Chinese characteristics. Manufacturers have greater pricing autonomy, with prices influenced by factors such as clinical value, innovation premium, competitive landscape, and brand strength. The entry of domestically developed alternatives to previously monopolized MNC products—such as HPV vaccines—has introduced competitive pricing pressure, making these technologies more accessible while still allowing for reasonable margins. The average import price of $3,744,281 per ton is largely generated in this segment, encompassing high-value products from MNCs that often face little or no direct local competition initially.
The historical trend in both import and export prices reveals a market experiencing inflationary pressure driven by innovation and input costs. The average import price grew by 5.8% in 2024, continuing a long-term trend of strong increase that included a period of exceptionally rapid growth. Similarly, the export price increased by 9.4% in 2024. These increases can be attributed to several factors: a changing product mix towards more expensive vaccines on both sides of trade, rising costs for research, raw materials, and compliant manufacturing, and general economic inflation. The peak in export prices observed in 2021 likely reflects pandemic-related demand and supply constraints for certain products.
Looking forward, price dynamics will be shaped by the interplay of several forces. Continued government pressure on healthcare costs may lead to expanded volume-based procurement for some high-expenditure Category II vaccines, a trend already observed in drug procurement. Simultaneously, successful innovation by domestic firms will create price competition in premium segments, potentially curbing the growth of ultra-high import prices. The overall trajectory suggests a gradual compression of the extreme value gap between imported and domestically consumed high-end vaccines, leading to a more normalized, yet still stratified, pricing environment as the market matures towards 2035.
Competitive Landscape
The competitive arena of the Chinese vaccine market is increasingly dynamic and segmented, featuring a clear but evolving distinction between multinational corporations (MNCs) and domestic players. MNCs, including Pfizer, GSK, Merck & Co., and Sanofi, maintain a dominant position in the high-value, innovative segment of the private market. They leverage global R&D pipelines, established brands, and sophisticated medical affairs capabilities to introduce novel vaccines, often enjoying a period of market exclusivity. Their strategies are focused on penetrating the Category II market, engaging with private healthcare providers, and navigating the complex market access and pricing environment.
Domestic competitors can be categorized into two main groups: established, large-scale manufacturers and emerging biotech innovators. The first group, led by state-affiliated giants like China National Biotec Group (CNBG) and Sinovac, commands the public NIP market through scale, cost advantage, and deep government relationships. Their portfolios are strong in traditional vaccines, and they are increasingly leveraging their commercial scale and capital to move into innovation. The second group, comprising companies like Walvax Biotechnology, CanSinoBIO, and Zhifei Biological, is more nimble and R&D-focused. These firms are responsible for many of the recent breakthrough domestic vaccines (e.g., HPV, COVID-19, pneumococcal) and are aggressively building pipelines in mRNA, recombinant proteins, and novel adjuvants.
The competitive strategies are diverging and converging in different areas. Key strategic thrusts include:
- Portfolio Diversification: Both MNCs and local firms are expanding their offerings to address lifecycle needs from pediatric to elderly populations, moving beyond single-product dependence.
- Technology Platform Investment: Heavy investment in mRNA, viral vector, and structure-based antigen design platforms is seen as critical for next-generation competitiveness.
- Partnerships and Collaborations: Strategic alliances are increasing, with MNCs licensing-in Chinese-developed technologies for global markets and domestic firms partnering with MNCs for co-promotion or to access complementary commercial capabilities.
- Market Access Agility: Success increasingly depends on expertise in navigating provincial procurement, private hospital tenders, and digital health platforms for consumer education and appointment scheduling.
The future competitive landscape to 2035 will be characterized by a blurring of the traditional MNC-domestic divide. Domestic champions will capture significant market share in several key innovative categories, transforming from volume suppliers to full-spectrum innovators. MNCs will continue to lead in areas of extreme scientific complexity but will face more potent competition. This will likely result in a more balanced, albeit fiercely competitive, market where success is determined by a combination of scientific innovation, operational excellence, commercial execution, and the ability to form strategic partnerships across the ecosystem.
Methodology and Data Notes
This report on the China Vaccines for Human Medicine market is built upon a robust and multi-faceted methodology designed to ensure analytical rigor, accuracy, and strategic relevance. The core of the research involves the systematic collection, cross-verification, and synthesis of data from a wide array of primary and secondary sources. Primary research includes interviews and surveys with key industry stakeholders, such as executives from vaccine manufacturers (both multinational and domestic), distributors, healthcare providers, procurement officials, and policy experts. These insights provide ground-level perspective on market dynamics, competitive strategies, and operational challenges.
Secondary research forms the quantitative backbone of the analysis, drawing from an extensive review of official and authoritative datasets. Key sources include trade statistics from China Customs and partner countries, production and consumption data from the National Bureau of Statistics and international bodies like the World Health Organization (WHO), and financial disclosures from publicly listed vaccine companies. Industry association reports, scientific publications, clinical trial registries, and regulatory announcements from the National Medical Products Administration (NMPA) are meticulously analyzed to track pipeline developments, approval trends, and policy shifts.
All market size, trade, and price figures presented, including the key metrics of consumption (13K tons), production (12K tons), and trade values and prices, are derived from this integrated data model for the base years. The model employs triangulation techniques to validate data points across different sources, ensuring consistency and reliability. Growth rates, market shares, and rankings are calculated based on these verified absolute figures. The forecast perspective through 2035 is developed using a combination of quantitative modeling—incorporating demographic projections, economic indicators, and historical trend analysis—and qualitative scenario planning based on identified drivers, constraints, and potential regulatory or technological disruptions.
It is critical to note the following data conventions and limitations: Market volumes (tons) are estimated based on standard dose equivalencies and may represent a proxy for overall market activity rather than literal weight. Value figures (USD) are primarily based on trade data and manufacturer sales, with adjustments made for distribution margins where possible. The report focuses on prophylactic vaccines for human use; therapeutic vaccines or animal vaccines are excluded from the scope. While every effort is made to ensure comprehensiveness, the fast-moving nature of the biopharmaceutical sector means some very recent developments may be incorporated qualitatively. This methodology provides a stable, auditable foundation for the strategic insights and conclusions presented throughout the report.
Outlook and Implications
The trajectory of the Chinese vaccine market from 2026 to 2035 points toward a period of maturation, consolidation, and value migration. The overarching theme will be the market's evolution from one defined by a stark dichotomy between high-value imports and volume-driven domestic production, towards a more integrated and sophisticated ecosystem where Chinese innovators become major players across the value spectrum. Domestic consumption will continue to grow, driven by an aging population, expansion of the NIP to include newer vaccines, and deepening penetration of Category II vaccines in lower-tier cities. However, growth rates in volume may moderate, while growth in market value will be sustained by the increasing share of innovative products.
On the supply side, the most significant implication is the substantial rise in China's innovation capacity. The domestic pipeline is expected to yield a steady stream of novel vaccines in areas such as oncology (therapeutic cancer vaccines), multi-valent pneumococcal, and next-generation respiratory virus vaccines. This will gradually alter import dependency, particularly for products where China achieves technological parity. MNCs will need to accelerate their own innovation cycles and consider more collaborative approaches, such as co-development deals with local partners or in-licensing Chinese innovations for global markets, to maintain their leadership positions in the face of heightened competition.
For stakeholders across the value chain, specific strategic implications are clear. Global suppliers must refine their China strategies beyond pure export models, potentially investing in local manufacturing partnerships for certain products and developing dedicated strategies for the evolving private and public procurement landscapes. Domestic manufacturers face the dual challenge of scaling their innovative offerings while optimizing costs for the competitive NIP tenders. They must also build global commercial capabilities to leverage their export potential beyond traditional emerging markets. Investors and policymakers will play a crucial role in funding the capital-intensive R&D and manufacturing scale-up required, while ensuring regulatory frameworks foster both innovation and safety.
In conclusion, the Chinese vaccine market stands at an inflection point. The decade to 2035 will see it solidify its position as the world's largest consumption market while dramatically ascending the global value chain in production and innovation. The resulting market will be larger, more valuable, and more complex. Success will require a nuanced understanding of the shifting balance between public health priorities and commercial dynamics, the evolving competitive landscape, and the intricate interplay between local innovation and global integration. This report provides the essential analysis to navigate this promising yet challenging future.
Frequently Asked Questions (FAQ) :
The countries with the highest volumes of consumption in 2024 were China, the United States and France, together accounting for 35% of global consumption. India, Poland, Spain, Germany, Japan, Russia and Indonesia lagged somewhat behind, together comprising a further 27%.
France remains the largest vaccine producing country worldwide, comprising approx. 33% of total volume. Moreover, vaccine production in France exceeded the figures recorded by the second-largest producer, the United States, twofold. The third position in this ranking was taken by China, with a 13% share.
In value terms, Ireland constituted the largest supplier of vaccines for human medicine to China, comprising 70% of total imports. The second position in the ranking was taken by the United States, with a 12% share of total imports. It was followed by France, with a 9.1% share.
In value terms, Morocco remains the key foreign market for vaccines for human medicine exports from China, comprising 22% of total exports. The second position in the ranking was taken by Pakistan, with a 9.5% share of total exports. It was followed by Egypt, with an 8.1% share.
The average vaccine export price stood at $1,367,562 per ton in 2024, picking up by 9.4% against the previous year. Overall, the export price showed a resilient increase. The most prominent rate of growth was recorded in 2020 when the average export price increased by 70% against the previous year. The export price peaked at $2,871,107 per ton in 2021; however, from 2022 to 2024, the export prices stood at a somewhat lower figure.
The average vaccine import price stood at $3,744,281 per ton in 2024, growing by 5.8% against the previous year. Over the period under review, the import price continues to indicate a strong increase. The pace of growth appeared the most rapid in 2017 an increase of 400%. The import price peaked in 2024 and is likely to continue growth in the near future.
This report provides a comprehensive view of the vaccines industry in China, tracking demand, supply, and trade flows across the national value chain. It explains how demand across key channels and end-use segments shapes consumption patterns, while also mapping the role of input availability, production efficiency, and regulatory standards on supply.
Beyond headline metrics, the study benchmarks prices, margins, and trade routes so you can see where value is created and how it moves between domestic suppliers and international partners. The analysis is designed to support strategic planning, market entry, portfolio prioritization, and risk management in the vaccines landscape in China.
Quick navigation
Key findings
- Domestic demand is shaped by both household and industrial usage, with trade flows linking local supply to imports and exports.
- Pricing dynamics reflect unit values, freight costs, exchange rates, and regulatory shifts that affect sourcing decisions.
- Supply depends on input availability and production efficiency, creating a distinct national cost curve.
- Market concentration varies by segment, creating different competitive landscapes and entry barriers.
- The 2035 outlook highlights where capacity investment and demand growth are most aligned within the country.
Report scope
The report combines market sizing with trade intelligence and price analytics for China. It covers both historical performance and the forward outlook to 2035, allowing you to compare cycles, structural shifts, and policy impacts.
- Market size and growth in value and volume terms
- Consumption structure by end-use segments
- Production capacity, output, and cost dynamics
- Trade flows, exporters, importers, and balances
- Price benchmarks, unit values, and margin signals
- Competitive context and market entry conditions
Product coverage
- Prodcom 21202145 - Vaccines for human medicine
Country coverage
Country profile and benchmarks
This report provides a consistent view of market size, trade balance, prices, and per-capita indicators for China. The profile highlights demand structure and trade position, enabling benchmarking against regional and global peers.
Methodology
The analysis is built on a multi-source framework that combines official statistics, trade records, company disclosures, and expert validation. Data are standardized, reconciled, and cross-checked to ensure consistency across time series.
- International trade data (exports, imports, and mirror statistics)
- National production and consumption statistics
- Company-level information from financial filings and public releases
- Price series and unit value benchmarks
- Analyst review, outlier checks, and time-series validation
All data are normalized to a common product definition and mapped to a consistent set of codes. This ensures that comparisons across time are aligned and actionable.
Forecasts to 2035
The forecast horizon extends to 2035 and is based on a structured model that links vaccines demand and supply to macroeconomic indicators, trade patterns, and sector-specific drivers. The model captures both cyclical and structural factors and reflects known policy and technology shifts in China.
- Historical baseline: 2012-2025
- Forecast horizon: 2026-2035
- Scenario-based sensitivity to income growth, substitution, and regulation
- Capacity and investment outlook for major producing companies
Each projection is built from national historical patterns and the broader regional context, allowing the report to show where growth is concentrated and where risks are elevated.
Price analysis and trade dynamics
Prices are analyzed in detail, including export and import unit values, regional spreads, and changes in trade costs. The report highlights how seasonality, freight rates, exchange rates, and supply disruptions influence pricing and margins.
- Price benchmarks by country and sub-region
- Export and import unit value trends
- Seasonality and calendar effects in trade flows
- Price outlook to 2035 under baseline assumptions
Profiles of market participants
Key producers, exporters, and distributors are profiled with a focus on their operational scale, geographic footprint, product mix, and market positioning. This helps identify competitive pressure points, partnership opportunities, and routes to differentiation.
- Business focus and production capabilities
- Geographic reach and distribution networks
- Cost structure and pricing strategy indicators
- Compliance, certification, and sustainability context
How to use this report
- Quantify domestic demand and identify the most attractive segments
- Evaluate export opportunities and prioritize target destinations
- Track price dynamics and protect margins
- Benchmark performance against leading competitors
- Build evidence-based forecasts for investment decisions
This report is designed for manufacturers, distributors, importers, wholesalers, investors, and advisors who need a clear, data-driven picture of vaccines dynamics in China.
FAQ
What is included in the vaccines market in China?
The market size aggregates consumption and trade data, presented in both value and volume terms.
How are the forecasts to 2035 built?
The projections combine historical trends with macroeconomic indicators, trade dynamics, and sector-specific drivers.
Does the report cover prices and margins?
Yes, it includes export and import unit values, regional spreads, and a pricing outlook to 2035.
Which benchmarks are included?
The report benchmarks market size, trade balance, prices, and per-capita indicators for China.
Can this report support market entry decisions?
Yes, it highlights demand hotspots, trade routes, pricing trends, and competitive context.